The current need to transition from non-renewable sources to green energy has prompted significant interest in efficient energy conversion. Methods that employ optimal thermal transport have accordingly become a focus of theoretical and experimental work within physics and nanotechnology.
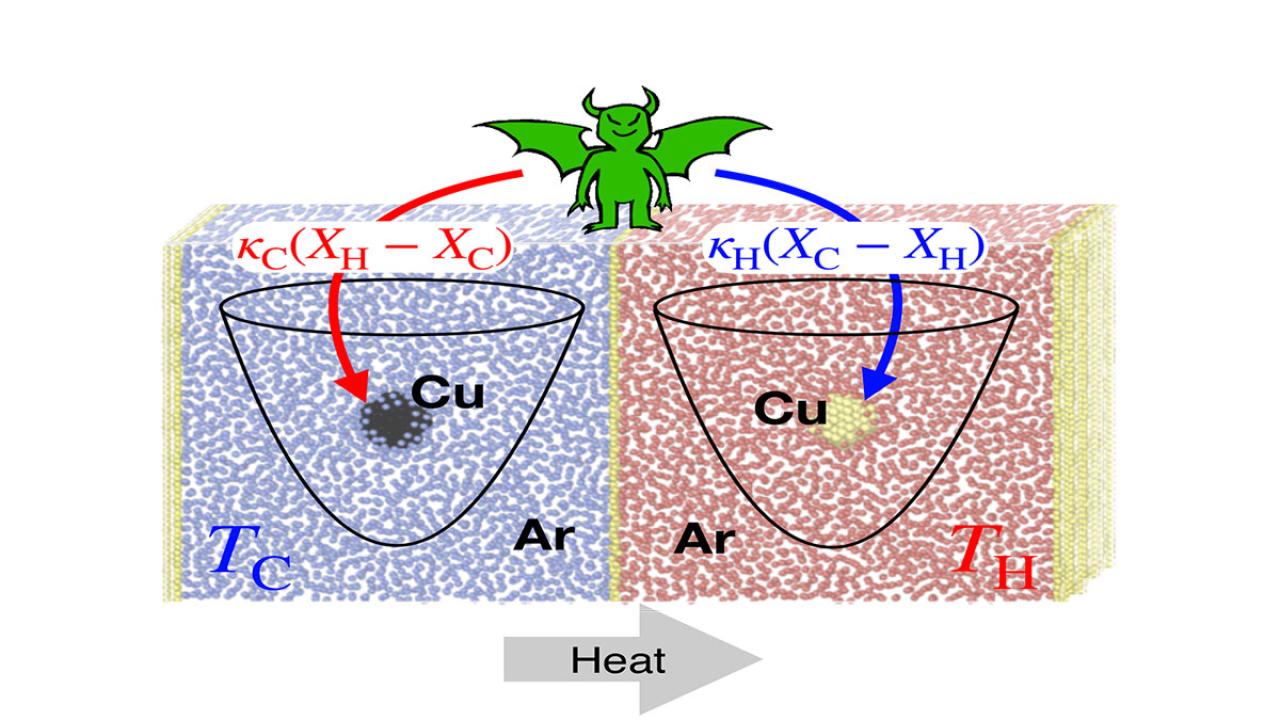
A new paper from ICTP researchers entitled "Nonreciprocal forces enable cold-to-hot heat transfer between nanoparticles” was recently published in Scientific Reports, an open-access journal from the Nature portfolio. ICTP researchers from the Quantitative Life Sciences (QLS) and Condensed Matter and Statistical Physics (CMSP) sections , in collaboration with an ICTP Associate, used a combination of Molecular Dynamics simulations and Stochastic Thermodynamics theoretical insights to describe heat transfer between two nanoparticles held at different temperatures, which interacted through nonreciprocal forces, i.e., forces that violate Newton's "action = reaction" 3rd law.
"Nonreciprocal forces is an exciting, emerging field within statistical physics,” explains QLS research scientist Edgar Roldan. “Researchers are just uncovering the tip of the iceberg within this topic, which has revealed intriguing and counterintuitive phenomena when these forces are present.”
"Fundamental laws in physics are all reciprocal,” adds Sarah Loos, a former QLS postdoc who is now a Marie Curie Fellow at the University of Cambridge. “However, such nonreciprocal interactions indeed emerge naturally as effective interactions in various complex systems, such as particles in flows, hot plasmas, and chemical systems. Furthermore, nonreciprocal interactions can be engineered, for example, by using closed-loop control. The proposed setup (two nonreciprocally coupled nanoparticles immersed in a fluid) is in principle realisable in state-of-the-art experiments with optical feedback."
The researchers discovered it is possible to construct a nano-refrigerator that generates a net cold-to-hot heat transfer at the expense of power exerted by these nonreciprocal forces. Their results show how nonreciprocal forces may be employed to quantify and control the direction and fluctuations of heat flow in natural and artificial nano-machines.
“We asked ourselves the question: is it possible to design a simple device at the nanoscale (made of just two nanoparticles) that can harvest heat from a cold thermal bath and transfer it to a thermal bath at a higher temperature? And if so, what are the design principles of such nano-machine?” says Roldan.
Most thermal machines, such as heat engines, extract energy in the form of heat from a hot bath (for example, in fuel burning). Part of this energy is transformed into useful work (exerted on the cylinders of the engine) while the rest is dissipated as heat in the colder external environment (the air). The opposite phenomenon occurs in refrigerators, which use work from the electrical power source to keep a sample cold. They extract heat from a cold bath (the inside of the refrigerator) which is dissipated as heat to the warmer external environment (the kitchen).
“We designed a tiny, nanoscale-sized refrigerator that works based on very simple principles,” adds Roldan. “One only needs nonreciprocal interactions between two nanoparticles to build such a refrigerator. We proposed this theoretically, made some predictions, and verified them using high-resolution molecular-dynamics simulations of silver nanoparticles immersed in argon. We then determined design principles for refrigerator devices. It is sufficient to exert linear (spring-like) nonreciprocal forces between particles.”
According to QLS associate Ali Rajabpour, “The main results of the study were the good matching between molecular dynamics simulations with statistical thermodynamic outputs for the design of a nano-refrigerator. Also, obtaining the spring constant ratio required to move from a direct heat transfer to reverse heat transfer.”
The project was conducted as part of an ongoing collaboration between CMSP and QLS at ICTP. Loos was the main author of the work, and developed most of the theoretical aspects employed using tools from stochastic thermodynamics. Molecular dynamics simulations of the silver nanoparticles in argon were performed by Saeed Arabha (now a PhD student at York University, Toronto) when he was a MSc student who visited the QLS and CMSP sections several times. Rajabpour, who was Arabha’s supervisor on the project, has been associated with both CMSP and QLS. CMSP research scientist Ali Hassanali supervised the molecular dynamics simulations, and Roldan proposed the project and supervised the theoretical calculations.
The study was part of a long-standing collaboration between Hassanali at CMSP and Roldan in QLS, which has led to the development of various successful projects at the interface between soft-matter biophysics and stochastic thermodynamics.
And as for the future? "We are further developing theoretical aspects to understand the role of nonreciprocal forces in thermodynamic machines,” says Roldan. "This is of utmost importance nowadays within the context of the energetic crisis, which has triggered an active push of current research towards clean and efficient energy-conversion devices.”
"The goal of continuing the work is to move towards more complex fluids and nanoparticles, as well as more accessible methods of applying nonreciprocal forces,” states Rajabpour.
"Meanwhile, researchers are starting to realise the significant role nonreciprocal interactions also play in various biological processes on a wide range of scales; from sub-cellular processes to the collective motion of many organisms,” adds Loos. "In this context, it will be very interesting to understand the implications of stochastic thermodynamic effects caused by nonreciprocal interactions, which we found in our study."